Abstract
Purpose
Considering the recent publication of the results of several clinical trials for metastatic clear cell renal cell carcinoma (mRCC), we performed a systematic review and meta-analysis of randomized studies comparing standard first-line VEGFR-targeted therapy to immune checkpoint inhibitors-based combinations for mRCC patients.
Methods
3960 patients from 5 randomized clinical trials where available for evaluation.
Result
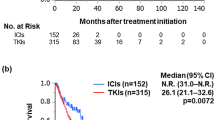
In the all-comers population, immunotherapy-based combinations were able to decrease the risk of death over the standard of care by 26% (HR 0.74; 95% CI 0.60–0.92; p = 0.006), to decrease the risk of progression by 21% (HR 0.79; 95% CI 0.72–0.86; p < 0.00001), and to increase the relative risk of response by 40% (HR 1.40; 95% CI 1.11–1.77; p = 0.006). For poor/intermediate-risk patients, the risk of death is decreased by 41% and the risk of progression by 27%.
Conclusions
The benefit of immunotherapy-based combinations in mRCC patients is independent from the IMDC risk group, but it is stronger for poor/intermediate-risk patients.





Similar content being viewed by others
References
Iacovelli R, Ciccarese C, Bria E, Bimbatti D, Fantinel E, Mosillo C, et al. Immunotherapy versus standard of care in metastatic renal cell carcinoma: a systematic review and meta-analysis. Cancer Treat Rev. 2018;70:112–7. https://doi.org/10.1016/j.ctrv.2018.08.007.
Rini BI, Plimack ER, Stus V, Gafanov R, Hawkins R, Nosov D, et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019. https://doi.org/10.1056/NEJMoa1816714.
Motzer RJ, Penkov K, Haanen J, Rini B, Albiges L, Campbell MT, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380(12):1103–15.
Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus Ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. 2018;378(14):1277–90.
Motzer RJ, Rini BI, McDermott DF, Arén Frontera O, Hammers HJ, Carducci MA, et al. Nivolumab plus ipilimumab versus sunitinib in first-line treatment for advanced renal cell carcinoma: extended follow-up of efficacy and safety results from a randomised, controlled, phase 3 trial. Lancet Oncol. 2019;20(10):1370–85.
Rini BI, Powles T, Atkins MB, Escudier B, McDermott DF, Suarez C, et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): a multicentre, open-label, phase 3, randomised controlled trial. Lancet. 2019;393(10189):2404–15.
McDermott DF, Huseni MA, Atkins MB, Motzer RJ, Rini BI, Escudier B, et al. Clinical activity and molecular correlates of response to atezolizumab alone or in combination with bevacizumab versus sunitinib in renal cell carcinoma. Nat Med. 2018;24(6):749–57.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535.
Mathes T, Kuss O. A comparison of methods for meta-analysis of a small number of studies with binary outcomes. Res Synth Methods. 2018;9(3):366–81.
Lueza B, Rotolo F, Bonastre J, Pignon JP, Michiels S. Bias and precision of methods for estimating the difference in restricted mean survival time from an individual patient data meta-analysis. BMC Med Res Methodol. 2016;16:37.
Heng DYC, Xie W, Regan MM, Harshman LC, Bjarnason GA, Vaishampayan UN, et al. External validation and comparison with other models of the International Metastatic Renal-Cell Carcinoma Database Consortium prognostic model: a population-based study. Lancet Oncol. 2013;14(2):141–8.
Funding
No funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Bersanelli received research funding by Roche, Pfizer, Seqirus, AstraZeneca, Bristol-Myers Squibb, Novartis, and Sanofi; she also received honoraria for advisory role and as speaker at scientific events by Bristol-Myers Squibb, Novartis, and Pfizer. Dr. Buti received honoraria for advisory role and as speaker at scientific events by Bristol-Myers Squibb, Novartis, IPSEN, and AstraZeneca. All other authors have no conflict of interest to declare.
Ethical approval
Ethical approval was not required.
Informed consent
Informed consent was not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Buti, S., Petrelli, F., Ghidini, A. et al. Immunotherapy-based combinations versus standard first-line treatment for metastatic clear cell renal cell carcinoma: a systematic review and meta-analysis. Clin Transl Oncol 22, 1657–1663 (2020). https://doi.org/10.1007/s12094-020-02292-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02292-z