Abstract
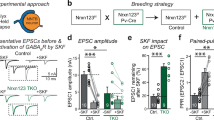
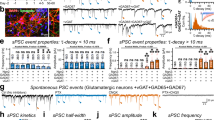
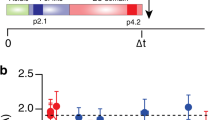
The differential formation of excitatory (glutamate-mediated) and inhibitory (GABA-mediated) synapses is a critical step for the proper functioning of the brain. An imbalance in these synapses may lead to various neurological disorders such as autism, schizophrenia, Tourette’s syndrome and epilepsy1,2,3,4. Synapses are formed through communication between the appropriate synaptic partners5,6,7,8. However, the molecular mechanisms that mediate the formation of specific synaptic types are not known. Here we show that two members of the fibroblast growth factor (FGF) family, FGF22 and FGF7, promote the organization of excitatory and inhibitory presynaptic terminals, respectively, as target-derived presynaptic organizers. FGF22 and FGF7 are expressed by CA3 pyramidal neurons in the hippocampus. The differentiation of excitatory or inhibitory nerve terminals on dendrites of CA3 pyramidal neurons is specifically impaired in mutants lacking FGF22 or FGF7. These presynaptic defects are rescued by postsynaptic expression of the appropriate FGF. FGF22-deficient mice are resistant to epileptic seizures, and FGF7-deficient mice are prone to them, as expected from the alterations in excitatory/inhibitory balance. Differential effects of FGF22 and FGF7 involve both their distinct synaptic localizations and their use of different signalling pathways. These results demonstrate that specific FGFs act as target-derived presynaptic organizers and help to organize specific presynaptic terminals in the mammalian brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rubenstein, J. L. & Merzenich, M. M. Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes Brain Behav. 2, 255–267 (2003)
Wassef, A., Baker, J. & Kochan, L. D. GABA and schizophrenia: a review of basic science and clinical studies. J. Clin. Psychopharmacol. 23, 601–640 (2003)
Singer, H. S. & Minzer, K. Neurobiology of Tourette’s syndrome: concepts of neuroanatomic localization and neurochemical abnormalities. Brain Dev. 25, S70–S84 (2003)
Möhler, H. GABAA receptors in central nervous system disease: anxiety, epilepsy, and insomnia. J. Recept. Signal Transduct. Res. 26, 731–740 (2006)
Sanes, J. R. & Lichtman, J. W. Development of the vertebrate neuromuscular junction. Annu. Rev. Neurosci. 22, 389–442 (1999)
Fox, M. A. & Umemori, H. Seeking long-term relationship: axon and target communicate to organize synaptic differentiation. J. Neurochem. 97, 1215–1231 (2006)
Waites, C. L., Craig, A. M. & Garner, C. C. Mechanisms of vertebrate synaptogenesis. Annu. Rev. Neurosci. 28, 251–274 (2005)
Dalva, M. B., McClelland, A. C. & Kayser, M. S. Cell adhesion molecules: signalling functions at the synapse. Nature Rev. Neurosci. 8, 206–220 (2007)
Umemori, H., Linhoff, M. W., Ornitz, D. M. & Sanes, J. R. FGF22 and its close relatives are presynaptic organizing molecules in the mammalian brain. Cell 118, 257–270 (2004)
Fox, M. A. et al. Distinct target-derived signals organize formation, maturation, and maintenance of motor nerve terminals. Cell 129, 179–193 (2007)
Guo, L., Degenstein, L. & Fuchs, E. Keratinocyte growth factor is required for hair development but not for wound healing. Genes Dev. 10, 165–175 (1996)
Steward, O. & Falk, P. M. Selective localization of polyribosomes beneath developing synapses: a quantitative analysis of the relationships between polyribosomes and developing synapses in the hippocampus and dentate gyrus. J. Comp. Neurol. 314, 545–557 (1991)
Danglot, L., Triller, A. & Marty, S. The development of hippocampal interneurons in rodents. Hippocampus 16, 1032–1060 (2006)
Gonzalez, A. M., Berry, M., Maher, P. A., Logan, A. & Baird, A. A comprehensive analysis of the distribution of FGF-2 and FGFR1 in the rat brain. Brain Res. 701, 201–226 (1995)
Zhang, X. et al. Receptor specificity of the fibroblast growth factor family. The complete mammalian FGF family. J. Biol. Chem. 281, 15694–15700 (2006)
Woodhams, P. L., Webb, M., Atkinson, D. J. & Seeley, P. J. A monoclonal antibody, Py, distinguishes different classes of hippocampal neurons. J. Neurosci. 9, 2170–2181 (1989)
Xiao, M. et al. Impaired hippocampal synaptic transmission and plasticity in mice lacking fibroblast growth factor 14. Mol. Cell. Neurosci. 34, 366–377 (2007)
Morimoto, K., Fahnestock, M. & Racine, R. J. Kindling and status epilepticus models of epilepsy: rewiring the brain. Prog. Neurobiol. 73, 1–60 (2004)
Racine, R. J. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr. Clin. Neurophysiol. 32, 281–294 (1972)
Umemori, H. & Sanes, J. R. Signal regulatory proteins (SIRPS) are secreted presynaptic organizing molecules. J. Biol. Chem. 283, 34053–34061 (2008)
Linhoff, M. W. et al. An unbiased expression screen for synaptogenic proteins identifies the LRRTM protein family as synaptic organizers. Neuron 61, 734–749 (2009)
Lin, Y. et al. Activity-dependent regulation of inhibitory synapse development by Npas4. Nature 455, 1198–1204 (2008)
Gibson, J. R., Huber, K. M. & Südhof, T. C. Neuroligin-2 deletion selectively decreases inhibitory synaptic transmission originating from fast-spiking but not from somatostatin-positive interneurons. J. Neurosci. 29, 13883–13897 (2009)
Schaeren-Wiemers, N. & Gerfin-Moser, A. A. single protocol to detect transcripts of various types and expression levels in neural tissue and cultured cells: in situ hybridization using digoxigenin-labelled cRNA probes. Histochemistry 100, 431–440 (1993)
Goslin, K., Asmussen, H. & Banker, G. in Culturing Nerve Cells (eds Banker, G. & Goslin, K.) 339–370 (MIT Press, 1998)
Acknowledgements
We thank J. Sanes and M. Hortsch for critical comments on the manuscript; M. Webb and P. Woodhams for the antibody Py; D. Sorenson for help with electron microscopy; A. Murayama for plasmid construction; M. De Freitas for help with histology; and M. Zhang for technical assistance. This work was supported by the Ester A. & Joseph Klingenstein Fund, the Edward Mallinckrodt Jr Foundation, the March of Dimes Foundation and the Whitehall Foundation (H.U.).
Author information
Authors and Affiliations
Contributions
A.T. and H.U. conceived and designed the experiments, performed or participated in each of the experiments and wrote the manuscript. E.M.J.-V. and M.A.S. performed the electrophysiological recordings. A.B.T. participated in the culture and histological experiments. D.J. performed the seizure-related experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-13 with legends and References. (PDF 3965 kb)
Rights and permissions
About this article
Cite this article
Terauchi, A., Johnson-Venkatesh, E., Toth, A. et al. Distinct FGFs promote differentiation of excitatory and inhibitory synapses. Nature 465, 783–787 (2010). https://doi.org/10.1038/nature09041
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09041
This article is cited by
-
Morphometric network-based abnormalities correlate with psychiatric comorbidities and gene expression in PCDH19-related developmental and epileptic encephalopathy
Translational Psychiatry (2024)
-
The FGF/FGFR system in the microglial neuroinflammation with Borrelia burgdorferi: likely intersectionality with other neurological conditions
Journal of Neuroinflammation (2023)
-
Fibroblast growth factor signaling in axons: from development to disease
Cell Communication and Signaling (2023)
-
Neonatal loss of FGFR2 in astroglial cells affects locomotion, sociability, working memory, and glia-neuron interactions in mice
Translational Psychiatry (2023)
-
Potentials of miR-9-5p in promoting epileptic seizure and improving survival of glioma patients
Acta Epileptologica (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.