Abstract
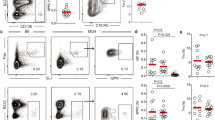
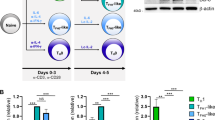
Interleukin 17 (IL-17) is a cytokine associated with inflammation, autoimmunity and defense against some bacteria. Here we show that IL-17 can promote autoimmune disease through a mechanism distinct from its proinflammatory effects. As compared with wild-type mice, autoimmune BXD2 mice express more IL-17 and show spontaneous development of germinal centers (GCs) before they increase production of pathogenic autoantibodies. We show that blocking IL-17 signaling disrupts CD4+ T cell and B cell interactions required for the formation of GCs and that mice lacking the IL-17 receptor have reduced GC B cell development and humoral responses. Production of IL-17 correlates with upregulated expression of the genes Rgs13 and Rgs16, which encode regulators of G-protein signaling, and results in suppression of the B cell chemotactic response to the chemokine CXCL12. These findings suggest a mechanism by which IL-17 drives autoimmune responses by promoting the formation of spontaneous GCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kolls, J.K. & Linden, A. Interleukin-17 family members and inflammation. Immunity 21, 467–476 (2004).
Lubberts, E., Koenders, M.I. & van den Berg, W.B. The role of T-cell interleukin-17 in conducting destructive arthritis: lessons from animal models. Arthritis Res. Ther. 7, 29–37 (2005).
Raza, K. et al. Early rheumatoid arthritis is characterized by a distinct and transient synovial fluid cytokine profile of T cell and stromal cell origin. Arthritis Res. Ther. 7, R784–R795 (2005).
Teunissen, M.B., Koomen, C.W., de Waal Malefyt, R., Wierenga, E.A. & Bos, J.D. Interleukin-17 and interferon-γ synergize in the enhancement of proinflammatory cytokine production by human keratinocytes. J. Invest. Dermatol. 111, 645–649 (1998).
Harrington, L.E. et al. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol. 6, 1123–1132 (2005).
Mangan, P.R. et al. Transforming growth factor-β induces development of the TH17 lineage. Nature 441, 231–234 (2006).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Komiyama, Y. et al. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J. Immunol. 177, 566–573 (2006).
Nakae, S., Nambu, A., Sudo, K. & Iwakura, Y. Suppression of immune induction of collagen-induced arthritis in IL-17–deficient mice. J. Immunol. 171, 6173–6177 (2003).
Sonderegger, I. et al. Neutralization of IL-17 by active vaccination inhibits IL-23–dependent autoimmune myocarditis. Eur. J. Immunol. 36, 2849–2856 (2006).
Taylor, B.A. et al. Genotyping new BXD recombinant inbred mouse strains and comparison of BXD and consensus maps. Mamm. Genome 10, 335–348 (1999).
Mountz, J.D. et al. Genetic segregation of spontaneous erosive arthritis and generalized autoimmune disease in the BXD2 recombinant inbred strain of mice. Scand. J. Immunol. 61, 128–138 (2005).
Wu, Y. et al. Synovial fibroblasts promote osteoclast formation by RANKL in a novel model of spontaneous erosive arthritis. Arthritis Rheum. 52, 3257–3268 (2005).
Hsu, H.C. et al. Production of a novel class of polyreactive pathogenic autoantibodies in BXD2 mice causes glomerulonephritis and arthritis. Arthritis Rheum. 54, 343–355 (2006).
Hsu, H.C. et al. Overexpression of activation-induced cytidine deaminase in B cells is associated with production of highly pathogenic autoantibodies. J. Immunol. 178, 5357–5365 (2007).
Allen, C.D. et al. Germinal center dark and light zone organization is mediated by CXCR4 and CXCR5. Nat. Immunol. 5, 943–952 (2004).
Allen, C.D., Okada, T., Tang, H.L. & Cyster, J.G. Imaging of germinal center selection events during affinity maturation. Science 315, 528–531 (2007).
Schwickert, T.A. et al. In vivo imaging of germinal centres reveals a dynamic open structure. Nature 446, 83–87 (2007).
Walker, L.S. et al. Established T cell–driven germinal center B cell proliferation is independent of CD28 signaling but is tightly regulated through CTLA-4. J. Immunol. 170, 91–98 (2003).
Cassese, G. et al. Inflamed kidneys of NZB/W mice are a major site for the homeostasis of plasma cells. Eur. J. Immunol. 31, 2726–2732 (2001).
Luzina, I.G. et al. Spontaneous formation of germinal centers in autoimmune mice. J. Leukoc. Biol. 70, 578–584 (2001).
Nordstrom, E., Abedi-Valugerdi, M. & Moller, E. Longevity of immune complexes and abnormal germinal centre formation in NZB mice. Scand. J. Immunol. 52, 477–482 (2000).
Wang, Y. & Carter, R.H. CD19 regulates B cell maturation, proliferation, and positive selection in the FDC zone of murine splenic germinal centers. Immunity 22, 749–761 (2005).
Berman, D.M., Wilkie, T.M. & Gilman, A.G. GAIP and RGS4 are GTPase-activating proteins for the Gi subfamily of G protein α subunits. Cell 86, 445–452 (1996).
Koelle, M.R. A new family of G-protein regulators—the RGS proteins. Curr. Opin. Cell Biol. 9, 143–147 (1997).
Watson, N., Linder, M.E., Druey, K.M., Kehrl, J.H. & Blumer, K.J. RGS family members: GTPase-activating proteins for heterotrimeric G-protein α-subunits. Nature 383, 172–175 (1996).
Estes, J.D. et al. Follicular dendritic cell regulation of CXCR4-mediated germinal center CD4 T cell migration. J. Immunol. 173, 6169–6178 (2004).
Moratz, C. et al. Regulator of G protein signaling 1 (RGS1) markedly impairs Gi α signaling responses of B lymphocytes. J. Immunol. 164, 1829–1838 (2000).
Shi, G.X., Harrison, K., Wilson, G.L., Moratz, C. & Kehrl, J.H. RGS13 regulates germinal center B lymphocytes responsiveness to CXC chemokine ligand (CXCL)12 and CXCL13. J. Immunol. 169, 2507–2515 (2002).
Armengol, M.P. et al. Chemokines determine local lymphoneogenesis and a reduction of circulating CXCR4+ T and CCR7 B and T lymphocytes in thyroid autoimmune diseases. J. Immunol. 170, 6320–6328 (2003).
Lubberts, E. et al. IL-17 promotes bone erosion in murine collagen-induced arthritis through loss of the receptor activator of NF-κB ligand/osteoprotegerin balance. J. Immunol. 170, 2655–2662 (2003).
Han, J.I., Huang, N.N., Kim, D.U. & Kehrl, J.H. RGS1 and RGS13 mRNA silencing in a human B lymphoma line enhances responsiveness to chemoattractants and impairs desensitization. J. Leukoc. Biol. 79, 1357–1368 (2006).
Moratz, C., Hayman, J.R., Gu, H. & Kehrl, J.H. Abnormal B-cell responses to chemokines, disturbed plasma cell localization, and distorted immune tissue architecture in Rgs1−/− mice. Mol. Cell. Biol. 24, 5767–5775 (2004).
Munthe, L.A., Corthay, A., Os, A., Zangani, M. & Bogen, B. Systemic autoimmune disease caused by autoreactive B cells that receive chronic help from Ig V region–specific T cells. J. Immunol. 175, 2391–2400 (2005).
Marion, T.N., Krishnan, M.R., Steeves, M.A. & Desai, D.D. Affinity maturation and autoimmunity to DNA. Curr. Dir. Autoimmun. 6, 123–153 (2003).
Fu, Y.X. & Storb, U. Immunology. Autoreactive B cells migrate into T cell territory. Science 297, 2006–2008 (2002).
William, J., Euler, C., Christensen, S. & Shlomchik, M.J. Evolution of autoantibody responses via somatic hypermutation outside of germinal centers. Science 297, 2066–2070 (2002).
Duan, B., Croker, B.P. & Morel, L. Lupus resistance is associated with marginal zone abnormalities in an NZM murine model. Lab. Invest. 87, 14–28 (2007).
Bave, U. et al. FcγRIIa is expressed on natural IFN-α–producing cells (plasmacytoid dendritic cells) and is required for the IFN-α production induced by apoptotic cells combined with lupus IgG. J. Immunol. 171, 3296–3302 (2003).
Meyers, J.A. et al. Blockade of TLR9 agonist-induced type I interferons promotes inflammatory cytokine IFN-γ and IL-17 secretion by activated human PBMC. Cytokine 35, 235–246 (2006).
Williams, R.W. et al. Genetic structure of the LXS panel of recombinant inbred mouse strains: a powerful resource for complex trait analysis. Mamm. Genome 15, 637–647 (2004).
Moratz, C., Harrison, K. & Kehrl, J.H. Regulation of chemokine-induced lymphocyte migration by RGS proteins. Methods Enzymol. 389, 15–32 (2004).
Schwarzenberger, P. et al. IL-17 stimulates granulopoiesis in mice: use of an alternate, novel gene therapy-derived method for in vivo evaluation of cytokines. J. Immunol. 161, 6383–6389 (1998).
Ye, P. et al. Requirement of interleukin 17 receptor signaling for lung CXC chemokine and granulocyte colony-stimulating factor expression, neutrophil recruitment, and host defense. J. Exp. Med. 194, 519–527 (2001).
Muramatsu, M. et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 102, 553–563 (2000).
Smith, K.G., Weiss, U., Rajewsky, K., Nossal, G.J. & Tarlinton, D.M. Bcl-2 increases memory B cell recruitment but does not perturb selection in germinal centers. Immunity 1, 803–813 (1994).
Acknowledgements
We thank Amgen for providing Il17r−/− B6 mice; E. Keyser and M.L. Spell (UAB FACS Core Facility) for operating the flow cytometry instrument; S. Williams (UAB High Resolution Imaging Facility) for acquiring confocal microscopic images; L. Harrington and C. Weaver for reagents and technical support in the TH-17 polarization experiments; F. Hunter for review of the manuscript; and C. Humber for secretarial assistance. This work was supported by the Arthritis Foundation (H.-C.H.), the American College of Rheumatology program (J.D.M. and R.H.C.), VA Merit Review Grants (J.D.M and R.H.C.), Daiichi-Sankyo (J.D.M.), and the National Institutes of Health (R24 DK64400 to R.G.L.).
Author information
Authors and Affiliations
Contributions
H.-C.H. and J.D.M. contributed to all studies; P.A.Y. was involved in all animal experiments, FACS staining, chemotactic experiments, and all qPCR and RT-PCR experiments; J.W., R.M., A.T., A.L.S., R.G.L. and R.H.C. contributed to all immunohistochemistry staining, imaging acquiring and imaging data interpretation; Q.W. contributed to animal experiments, FACS staining, chemotactic experiments, and ELISA and ELISPOT analyses; J.C. contributed to FACS analysis and adenovirus propagation; J.Y. contributed to T cell polarization experiments and chemotactic experiments; J.K.K. contributed to the generation of AdIL-17 and AdIL-17R:Fc; T.G., H.X. and R.W.W. contributed to the generation of Il17r−/− BXD2 and GFP+ BXD2 mice; T.-v.L.L. and D.D.C. contributed to the studies of SHM analysis.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Tables 1–3 and Methods (PDF 322 kb)
Rights and permissions
About this article
Cite this article
Hsu, HC., Yang, P., Wang, J. et al. Interleukin 17–producing T helper cells and interleukin 17 orchestrate autoreactive germinal center development in autoimmune BXD2 mice. Nat Immunol 9, 166–175 (2008). https://doi.org/10.1038/ni1552
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1552
This article is cited by
-
RORγt inverse agonist TF-S14 inhibits Th17 cytokines and prolongs skin allograft survival in sensitized mice
Communications Biology (2024)
-
Blocking interleukin-23 ameliorates neuromuscular and thymic defects in myasthenia gravis
Journal of Neuroinflammation (2023)
-
Modulation of Mesenchymal Stem Cells-Mediated Adaptive Immune Effectors’ Repertoire in the Recovery of Systemic Lupus Erythematosus
Stem Cell Reviews and Reports (2023)
-
Norcantharidin ameliorates the development of murine lupus via inhibiting the generation of IL-17 producing cells
Acta Pharmacologica Sinica (2022)
-
Increased Expression of CD95 in CD4+ Effector Memory T Cells Promotes Th17 Response in Patients with Myasthenia Gravis
Journal of Neuroimmune Pharmacology (2022)