Abstract
Background
Currently, complete lymph node dissection (CLND) is recommended after identification of a metastatic lymph node by sentinel lymph node biopsy (SLNB). Guidelines suggest that CLND should be performed as a separate procedure, and a sufficient number of nodes should be examined. Our objective was to examine the utilization, timing, and adequacy of CLND for melanoma in the United States.
Methods
From the National Cancer Data Base, patients diagnosed with stage I to III melanoma during 2004–2005 were identified. Multiple logistic regression was used to assess factors associated with CLND utilization, timing (separate operation from SLNB), and adequacy (examination of ≥10 nodes).
Results
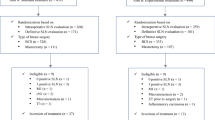
Of the 44,548 patients identified, 47.5% were pathologic stage IA, 23.8% stage IB, 14.1% stage II, and 14.6% stage III. Of the 17% (2942 of 17,524) with nodal metastases on SLNB, only 50% underwent a CLND. Patients were significantly less likely to undergo a CLND after SLNB if >75 years old or had lower extremity melanomas. Of the patients who underwent a CLND, only 42% underwent the CLND at a separate procedure after the SLNB. Of those who underwent a CLND, 69.2% had ≥10 nodes examined. Patients were significantly less likely to have ≥10 nodes examined if they were >75 years old or had lower extremity melanomas. Patients treated at NCCN/NCI-designated centers were significantly more likely to undergo nodal evaluation in concordance with established guidelines.
Conclusions
Only half of patients with sentinel node-positive melanoma underwent CLND. Quality surveillance measures are needed to monitor, standardize, and improve the care of patients with malignant melanoma.

Similar content being viewed by others
References
Desmond RA, Soong SJ. Epidemiology of malignant melanoma. Surg Clin North Am 2003; 83:1–29.
Jemal A, Siegel R, Ward E, et al. Cancer statistics. CA Cancer J Clin 2007; 57:43–66
Horm JW, Sondik EJ. Person-years of life lost due to cancer in the United States, 1970 and 1984. Am J Public Health 1989; 79:1490–3
Gershenwald JE, Thompson W, Mansfield PF, et al. Multi-institutional melanoma lymphatic mapping experience: the prognostic value of sentinel lymph node status in 612 stage I or II melanoma patients. J Clin Oncol 1999; 17:976–83
Morton DL, Wen DR, Wong JH, et al. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch Surg 1992; 127:392–9
Balch CM, Soong S, Ross MI, et al. Long-term results of a multi-institutional randomized trial comparing prognostic factors and surgical results for intermediate thickness melanomas (1.0 to 4.0 mm). Intergroup Melanoma Surgical Trial. Ann Surg Oncol 2000; 7:87–97
Morton DL, Thompson JF, Cochran AJ, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med 2006; 355:1307–17
Morton DL, Scheri RP, Balch CM. Can completion lymph node dissection be avoided for a positive sentinel node in melanoma? Ann Surg Oncol 2007; 14:2437–9
Wong SL, Morton DL, Thompson JF, et al. Melanoma patients with positive sentinel nodes who did not undergo completion lymphadenectomy: a multi-institutional study. Ann Surg Oncol 2006; 13:809–16
Complete lymph node dissection or observation in treating patients with localized melanoma and sentinel node metastasis who have undergone sentinel lymphadenectomy (MSLT-II). Available at: http://www.clinicaltrials.gov/ct/show/NCT00389571?order=1. Accessed March 14, 2008
National Comprehensive Cancer Network (NCCN) clinical practice guidelines in oncology: melanoma. v.2.2007. Available at: http://www.nccn.org/. Accessed March 14, 2008
Bilimoria KY, Stewart AK, Winchester DP, et al. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol 2008; 15:683–90
Winchester DP, Stewart AK, Bura C, et al. The National Cancer Data Base: a clinical surveillance and quality improvement tool. J Surg Oncol 2004; 85:1–3
World Health Organization. International Classification of Disease for Oncology. 3rd ed. Geneva: World Health Organization, 2000
Department of Health, Human Services. The International Classification of Diseases. 9th revised. clinical modification: ICD-9-CM. Washington, DC: Government Printing Office, 1998
Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40:373–83
Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol 1992; 45:613–9
United States Census Bureau. Census 2000. http://www.census.gov/main/www/cen2000.html. Accessed March 14, 2008
Facility Oncology Registry Data Standards. Chicago: Commission on Cancer, 2004
AJCC Cancer Staging Manual, 6th ed. Chicago IL: Springer, 2002
Panageas KS, Schrag D, Riedel E, et al. The effect of clustering of outcomes on the association of procedure volume and surgical outcomes. Ann Intern Med 2003; 139:658–65
Hosmer J, Lemeshow S. Applied logistic regression. New York: John Wiley & Sons, 1999
Cormier JN, Xing Y, Ding M, et al. Population-based assessment of surgical treatment trends for patients with melanoma in the era of sentinel lymph node biopsy. J Clin Oncol 2005; 23:6054–62
Wong SL, Brady MS, Busam KJ, et al. Results of sentinel lymph node biopsy in patients with thin melanoma. Ann Surg Oncol 2006; 13:302–9
Sabel MS, Griffith K, Sondak VK, et al. Predictors of nonsentinel lymph node positivity in patients with a positive sentinel node for melanoma. J Am Coll Surg 2005; 201:37–47
Scheri RP, Essner R, Turner RR, et al. Isolated tumor cells in the sentinel node affect long-term prognosis of patients with melanoma. Ann Surg Oncol 2007; 14:2861–6
Lee JH, Essner R, Torisu-Itakura H, et al. Factors predictive of tumor-positive nonsentinel lymph nodes after tumor-positive sentinel lymph node dissection for melanoma. J Clin Oncol 2004; 22:3677–84
Wrightson WR, Wong SL, Edwards MJ, et al. Complications associated with sentinel lymph node biopsy for melanoma. Ann Surg Oncol 2003; 10:676–80
Baas PC, Schraffordt Koops H, Hoekstra HJ, et al. Groin dissection in the treatment of lower-extremity melanoma. Short-term and long-term morbidity. Arch Surg 1992; 127:281–6
Bland KI, Klamer TW, Polk HC Jr, et al. Isolated regional lymph node dissection: morbidity, mortality and economic considerations. Ann Surg 1981; 193:372–6
Hughes TM, Thomas JM. Combined inguinal and pelvic lymph node dissection for stage III melanoma. Br J Surg 1999; 86:1493–8
Cascinelli N, Bombardieri E, Bufalino R, et al. Sentinel and nonsentinel node status in stage IB and II melanoma patients: two-step prognostic indicators of survival. J Clin Oncol 2006; 24:4464–71
Kirkwood JM, Ibrahim JG, Sosman JA, et al. High-dose interferon alfa-2b significantly prolongs relapse-free and overall survival compared with the GM2-KLH/QS-21 vaccine in patients with resected stage IIB-III melanoma: results of intergroup trial E1694/S9512/C509801. J Clin Oncol 2001; 19:2370–80
Kirkwood JM, Strawderman MH, Ernstoff MS, et al. Interferon alfa-2b adjuvant therapy of high-risk resected cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J Clin Oncol 1996; 14:7–17
Starz H, Siedlecki K, Balda BR. Sentinel lymphonodectomy and s-classification: a successful strategy for better prediction and improvement of outcome of melanoma. Ann Surg Oncol 2004; 11(3 Suppl):162S–8S
Bostick P, Essner R, Glass E, et al. Comparison of blue dye and probe-assisted intraoperative lymphatic mapping in melanoma to identify sentinel nodes in 100 lymphatic basins. Arch Surg 1999; 134:43–9
Malin JL, Kahn KL, Adams J, et al. Validity of cancer registry data for measuring the quality of breast cancer care. J Natl Cancer Inst 2002; 94:835–44
Yu LL, Flotte TJ, Tanabe KK, et al. Detection of microscopic melanoma metastases in sentinel lymph nodes. Cancer 1999; 86:617–27
Stojadinovic A, Allen PJ, Clary BM, et al. Value of frozen-section analysis of sentinel lymph nodes for primary cutaneous malignant melanoma. Ann Surg 2002; 235:92–8
Tanis PJ, Boom RP, Koops HS, et al. Frozen section investigation of the sentinel node in malignant melanoma and breast cancer. Ann Surg Oncol 2001; 8:222–6
Galliot-Repkat C, Cailliod R, Trost O, et al. The prognostic impact of the extent of lymph node dissection in patients with stage III melanoma. Eur J Surg Oncol 2006; 32:790–4
Baxter NN, Tuttle TM. Inadequacy of lymph node staging in gastric cancer patients: a population-based study. Ann Surg Oncol 2005; 12:981–7
Baxter NN, Virnig DJ, Rothenberger DA, et al. Lymph node evaluation in colorectal cancer patients: a population-based study. J Natl Cancer Inst 2005; 97:219–25
Schwarz RE, Smith DD. Extent of lymph node retrieval and pancreatic cancer survival: information from a large US population database. Ann Surg Oncol 2006; 13:1189–200
Bilimoria KY, Palis BE, Stewart AK, et al. Impact of tumor location on nodal evaluation for colon cancer. Dis Colon Rectum 2008; 51:154–61
Bilimoria KY, Talamonti MS, Tomlinson JS, et al. Impact of hospital type and volume on lymph node evaluation for gastric and pancreatic cancer. Arch Surg (in press)
McGlynn EA, Asch SM, Adams J, et al. The quality of health care delivered to adults in the United States. N Engl J Med 2003; 348:2635–45
Acknowledgment
Supported by the American College of Surgeons Clinical Scholars in Residence program (K.Y.B.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bilimoria, K.Y., Balch, C.M., Bentrem, D.J. et al. Complete Lymph Node Dissection for Sentinel Node-Positive Melanoma: Assessment of Practice Patterns in the United States. Ann Surg Oncol 15, 1566–1576 (2008). https://doi.org/10.1245/s10434-008-9885-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-008-9885-2