Abstract
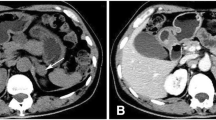
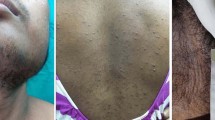
A 45-year-old man presented with gynecomastia, hypertension and a large left adrenal mass. Further evaluation revealed elevated serum concentrations of estrogen, estrone sulfate, androstenedione, dehydroepiandrosterone, dehydroepiandrosterone sulfate, deoxycorticosterone, and aldosterone and increased 24-hour urinary 17-ketosteroid and free cortisol excretion. Removal of a 10 kg adrenocortical carcinoma led to normalization of the hormone concentrations and partial resolution of the gynecomastia. There was no clinical evidence of metastases. Incubation of tumor slices demonstrated that the tumor had an active aromatase and sulfotransferase. We estimated that about half the serum estrone arose from peripheral conversion of androstenedione. Feminizing adrenocortical carcinomas are rare and this case is unusual given the lack of clinical metastases and the probable dual source of estrogen from tumor as well as from the peripheral conversion of tumor-derived androgens.
Similar content being viewed by others
References
Gabrilove J.L., Sharma D.C., Wotiz H.H., Dorfman R.I. Feminizing adrenal cortical tumors in the male: a review of 52 cases including a case report. Medicine 44: 37, 1965.
Gabrilove J.L., Nicolis G.L., Hausknecht R.V., Wotiz H.H. Feminizing adrenocortical carcinoma in a man. Cancer 25: 153, 1970.
Golder M., Millington D., Cowley T., Griffiths K., Roberts H., London D.R., Butt W.R. Hormonal control of steroid synthesis by a feminizing adrenal tumor. J. Endocrinol. 67: 17p, 1975.
Veldhuis J., Sowers J., Rogol A., Klein F.A., Miller N., Dufau M.L. Pathophysiology of male hypogonadism associated with endogenous hyperestrogenism. N. Engl. J. Med. 312: 1371, 1985.
Fang V.S. Establishment and characterization of a strain of human adrenal tumor cells that secrete estrogen. Proc. Natl. Acad. Sci. USA 74: 1067, 1977.
Mathur R.S., Williamson H.O., Moody L.O., Diczfalusy E. In-vitro sterol and steroid biogenesis by a feminizing adrenocortical carcinoma. Acta Endocrinol. (Copenh.) 73: 518, 1973.
McKenna T.J., O’Connell Y., Cunningham S., McCabe M., Culliton M. Steroidogenesis in an estrogen-producing adrenal tumor in a young woman: comparison with steroid profiles associated with cortisol- and androgen-producing tumors. J. Clin. Endocrinol. Metab. 70: 28, 1990.
Orczyk G.P., Mordes J.R., Longcope C. Aromatase activity in a rat Leydig cell tumor. Endocrinology 120: 1482, 1987.
Longcope C. The metabolism of estrone sulfate in normal males. J. Clin. Endocrinol. Metab. 34: 113, 1972.
Freeman D. Steroid hormone-producing tumors in man. Endocr. Rev. 7: 204, 1986.
Baird D.T., Uno A., Melby J.C. Adrenal secretion of androgens and oestrogens. J. Endocrinol. 45: 135, 1969.
Longcope C. Adrenal and gonadal androgen secretion in normal females. Clin. Endocrinol. Metabol. 15: 213, 1986.
Longcope C., Kato T., Horton R. Conversion of blood androgens to estrogens in normal adult men and women. J. Clin. Invest. 48: 2191, 1969.
Longcope C. Methods and results of aromatization studies in-vivo. Cancer Res. 42: 3307S, 1982.
Baird D., Horton R., Longcope C., Tait J.F. Steroid prehormones. Pros. Biol. Med. 11: 383, 1968.
Longcope C. Peripheal aromatization: studies on controlling factors. Steroids 50: 253, 1987.
Longcope C., Bourget C., Flood C. The production and aromatization of dehydroepiandrosterone in postmenopausal women. Maturitas 4: 325, 1982.
Haning R. Jr., Carleson I., Flood C., Hackett R., Longcope C. Metabolism of dehydroepiandrosterone sulfate (DS) in normal women and women with high DS concentrations. J. Clin. Endocrinol. Metab. 73: 1210, 1991.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zayed, A., Stock, J.L., Liepman, M.K. et al. Feminization as a result of both peripheral conversion of androgens and direct estrogen production from an adrenocortical carcinoma. J Endocrinol Invest 17, 275–278 (1994). https://doi.org/10.1007/BF03348975
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03348975